5 Steps to Draw Lewis Structures
Before learning techniques about how to draw Lewis structures, you must first understand what Lewis structures are and why they are important. A Lewis structure is a pictorial diagram showing how electrons get distributed around an atom. Lewis structures are drawn to help one understand or predict how many types of bonds can be formed around a given atom. The drawing is started by determining types of covalent bonds that are formed after combining atoms. These types of covalent bonds are dependent on their valence electron numbers. The final structure of a Lewis structure is achieved when all elements (except hydrogen atoms) get eight (octet) valence electrons. Finally, all this information helps in the prediction of molecular geometry.
How to Draw Lewis Structures
For those who are concerned about how to draw Lewis structures, below is a simple procedure on how to do it.
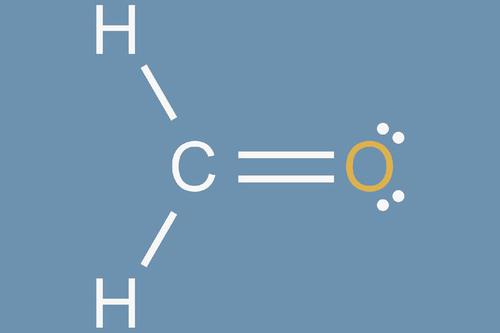
1. Count the Total Number of Valence Electrons
The first step is determining the count of valence electrons found in all the atoms that make the molecule you are dealing with. To find the number of valence electrons, you will have to do a simple computation.
Here is an example. If you are planning to draw a carbon tetrachloride (CCI4) Lewis structure, you will have to count all the valence electrons involved in each part of the overall molecule.
Because of the formula of the CCl4 molecule, you can tell the carbon atom has four valence electrons. Secondly, you will add seven valence electrons for every chlorine atom CCl4 molecule. Finally, because carbon tetrachloride is not charged, you will not add an electron for that component of the molecule. Thus the CCl4 valence electron computation will give you the following equation:
4 + (47) = 32. The total electron number is 32. To review, the first four came from the carbon atom's four valence electrons, and the 47 came from the four chlorine atoms which each have seven valence electrons.
2. Put the Least Electronegative Atom in the Center
We have found the total number of valence electrons. Now, as we continue to review how to draw Lewis structures, we must decide which atom will be drawn in the center of our Lewis structure. The atom with the least electronegative charge should be put at the center when drawing. (It has the most available "real estate” around it, so it works well to draw the least-electronegative first. This way you will have space around to draw the rest of the atoms.)
How do you determine the electronegativity of an atom? Use the periodic table of elements or an electronegativity list. As you read the periodic table, electronegativity decreases as you go down the groups while it increases as you read each row from left to right.
After placing the least electronegative atom at the center, the other atoms are to be drawn or placed around it.
In the case of our example atom, CCl4, you should draw the Carbon atom in the center. Carbon has four valence electrons, while Chlorine has seven valence electrons.
3. Draw a Skeletal Structure
When drawing this skeletal structure, remember that the atom with the most available bonding sites (remember, it is the atom that has the smallest electronegative charge--the atom that you identified from the previous instruction) is to remain at the center. In other words, the atom which can allow the most other atoms get bonded to it should be centrally placed on the skeletal structure. A centrally positioned atom can allow up to four connections around it.
So, returning to our example of the CCl4, you will draw the Carbon at the center. Since the carbon atom will need to add 4 more atoms to achieve its desired 8 (octet) electrons, it will bond with each of the four chlorine atoms. These chlorine atoms also will want to achieve 8 (octet) electrons each.Thus, the total octet electrons will then be 8 + (4 * 8) = 40. (The 8 in this equation is for the carbon, while the (4 * 8) represents the 4 chlorine atoms each seeking 8 electrons.
Now, we have another computation. We just found the total electrons of a carbon atom and four chlorine atoms--40--but we have to use this number along with the total number of valence electrons in a simple equation. Take your total number of electrons, which is 40, and subtract 32--the number of valence electrons. The difference is 8.
Now, using your chemistry knowledge that a covalent bond needs to share a pair of electrons to make a strong enough bond, you will divide 8 (the number of valence electrons) by 2 (the number of electrons in each strong bond). The quotient is 4--which is the number of chemical bonds to draw around your central carbon atom.
4. Place Electrons Around Outside Atoms
At this stage, you must apply what you know about how to do Lewis dot structure. The diagram must be completed, showing the last of the electrons in the form of dots on the outer atoms bonded to the central atom. You may get these dots wrong on your first few tries, but you will learn with each attempt. Remember to follow the steps: knowing the number of valence electrons, placing the electron with the lowest electronegative charge at the center, drawing each remaining atom around it, and noting the electron bonds between the central atom and each remaining atom. Finally, each outer atom will get two electrons, which are symbolized by dots on the drawing.
5. Place Remaining Electrons Around the Central Atom
This is the last Lewis structure step. After you have placed double electrons on all the outer atoms, the skeletal structure is completed by placing the remaining electrons next to the central atom, rounding out its octet. Each bond that was created in step C will be represented by a straight line--these lines should come in pairs to represent the double bonds necessary for strong covalent bonds. After you count up all the solid lines representing lone pairs and all the extra dots representing atoms that complete octets, then double-check your math. If there are more electrons placed around the center atom than its standard octet, then the arithmetic from step A (counting the valence electrons) might be in error.
Lewis structures are useful. They provide a visual diagram of the molecular bonds, light elements, and oxidation states you are studying in your chemistry classes. There are some very particular details to master while learning to draw Lewis structures, but once you understand the procedure, they will help you master the details of your understanding of chemical bonds.
YOU MAY LIKE
-
Disadvantages and Advantages to Keep Animals in Zoos
-
Do You Know Why People Don’t Read Books?
-
Most Interesting Challenges to Do With Your Friends
-
3 Types of Elements on the Periodic Table and Their Properties
-
What Are the 3 Parts of a Nucleotide and Nucleotide Examples
-
Tips on How to Write a “Thank You” E-mail
-
11 Amazing Chemical Reactions in Everyday Life!
-
The Significance of Knowing Your Strengths and Weaknesses
-
Different Truth or Dare Questions for Adults
-
DNA VS RNA: A Comparative Study
